
All News
2 articles in total
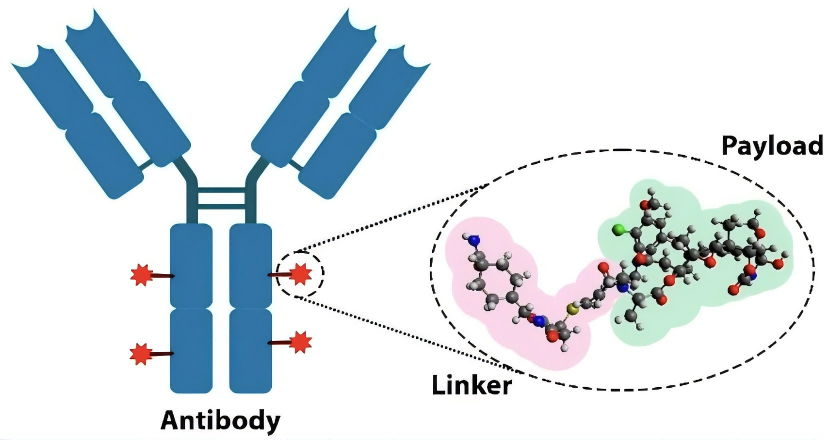
Linker Design in Antibody-Drug Conjugates: Strategies and Cutting-Edge Advances
Antibody-drug conjugates (ADCs) enable targeted cancer therapy by linking antibodies to cytotoxic drugs, realizing Paul Ehrlich’s “magic bullet.” A recent review highlights linkers as key to stability and efficacy: cleavable linkers (e.g., enzyme-sensitive Val-Cit) enable precise tumor release, while non-cleavable linkers offer better plasma stability. Core strategies include site-specific conjugation, release triggers, and hydrophilic spacers to prevent aggregation. Advances feature dual-stimuli triggers, traceless release, branched structures, and AI tools like Linker-GPT. With 17 ADCs approved, challenges remain in immunogenicity, manufacturing, and resistance; future efforts focus on computational modeling and nanotechnology to expand applications beyond oncology.
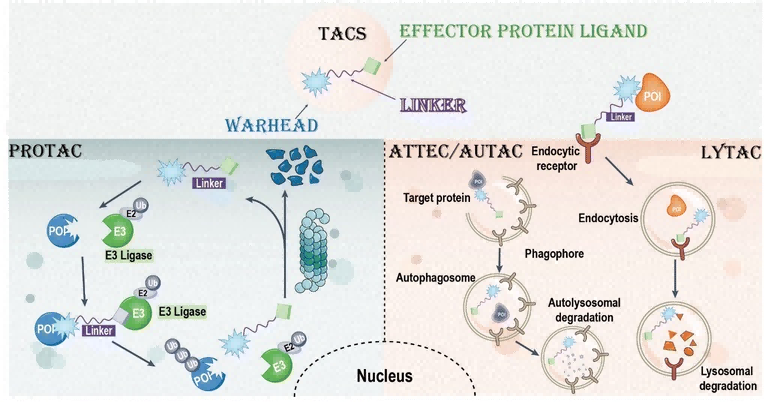
Review of the Development History of TACs Multifunctional Linkers
Targeted chimeras (TACs, such as PROTACs and LYTACs) represent a potential strategy for the selective degradation of difficult-to-drug proteins, acting through bifunctional molecules or molecular glues to provide novel therapies for diseases that are difficult to treat with traditional small molecules. As a bridge connecting target protein ligands and effector protein ligands, the linker of TACs is crucial to molecular conformation and activity. Recent advancements in photo-controlled and cleavable linker design have improved the efficiency and spatiotemporal control of TACs, but challenges remain, such as optimizing stability.
